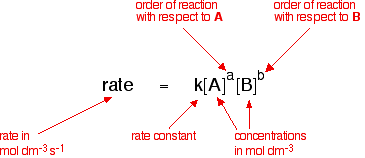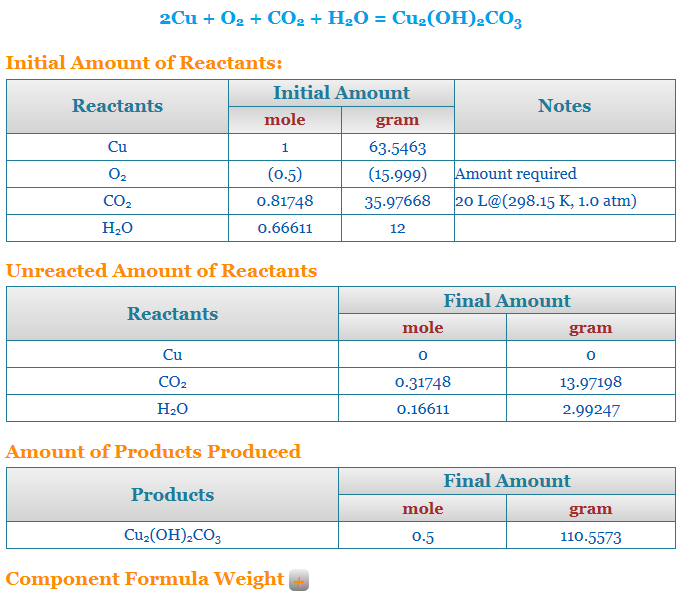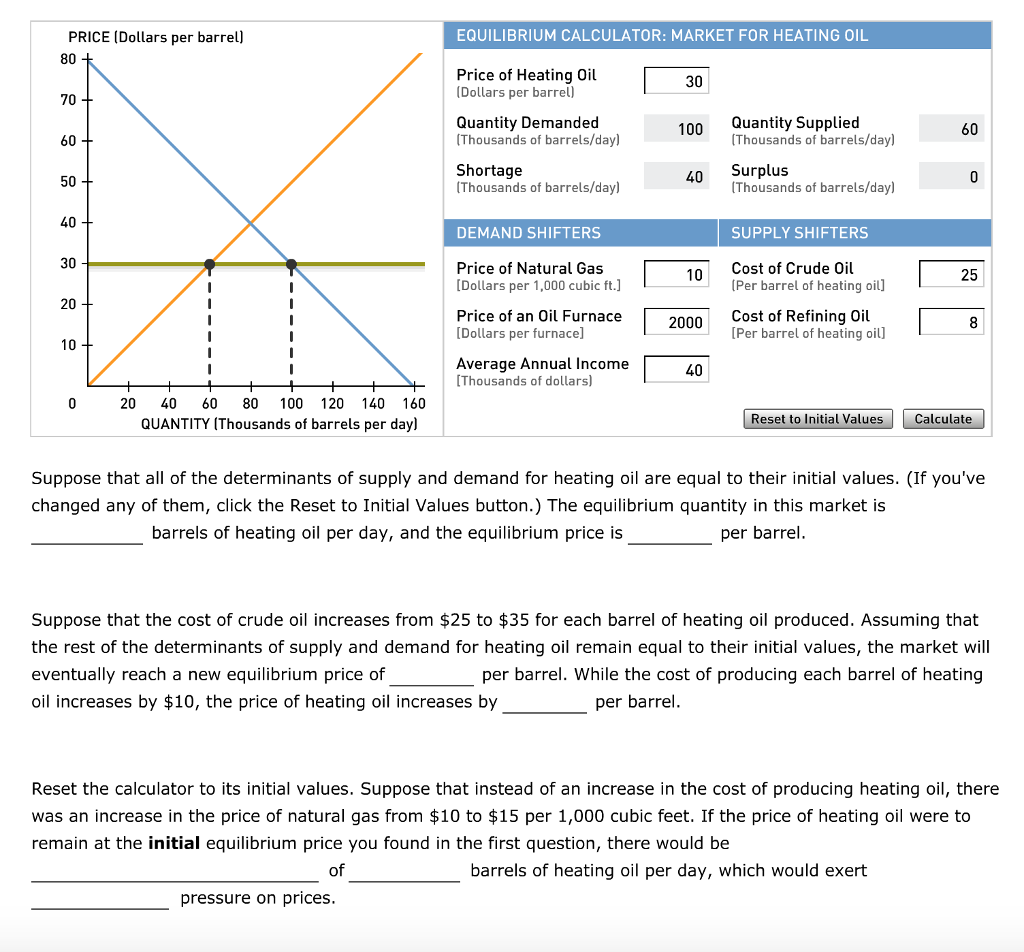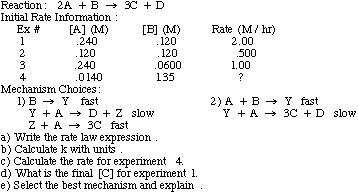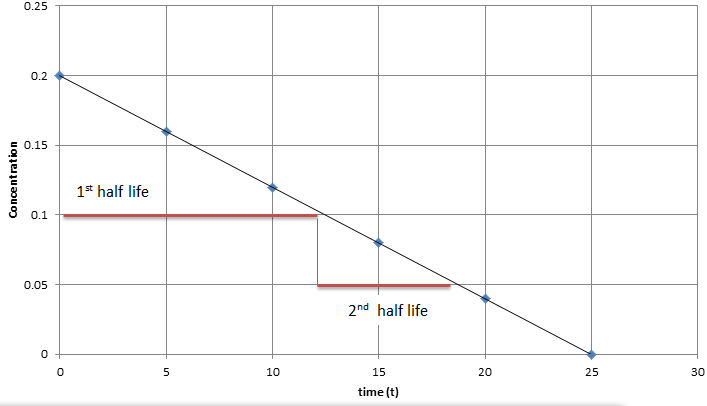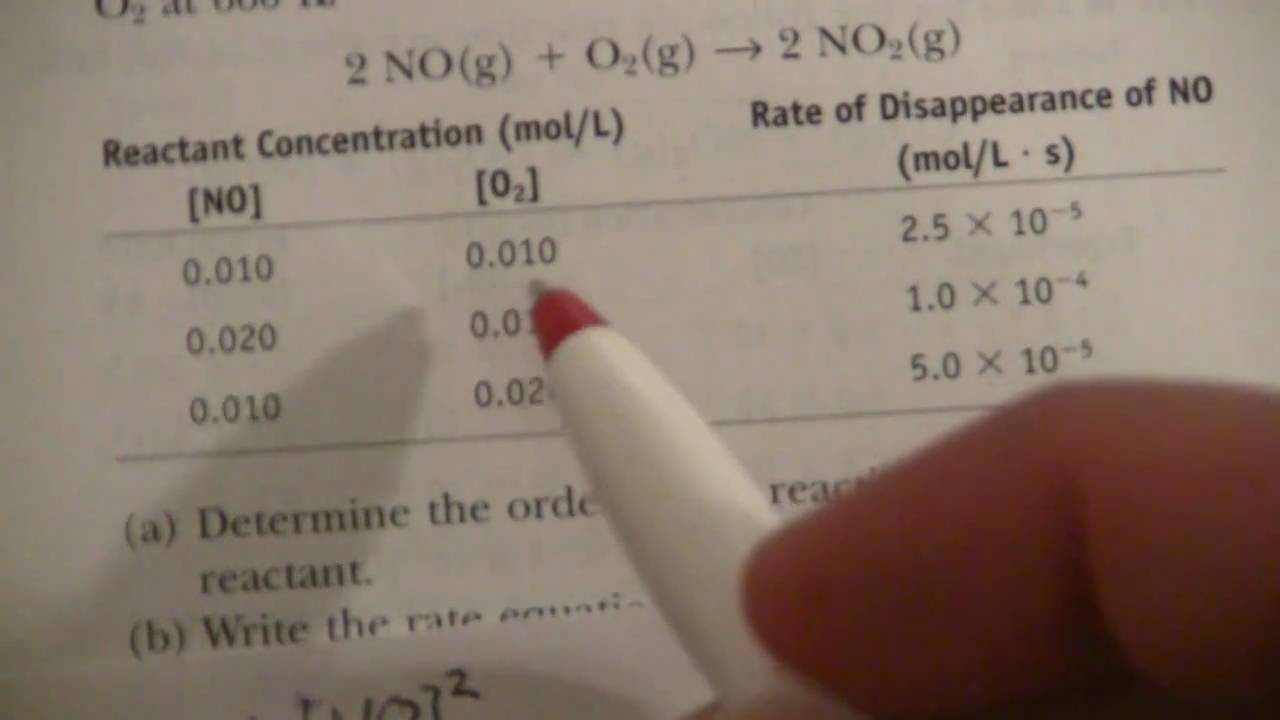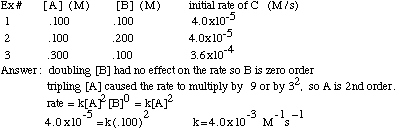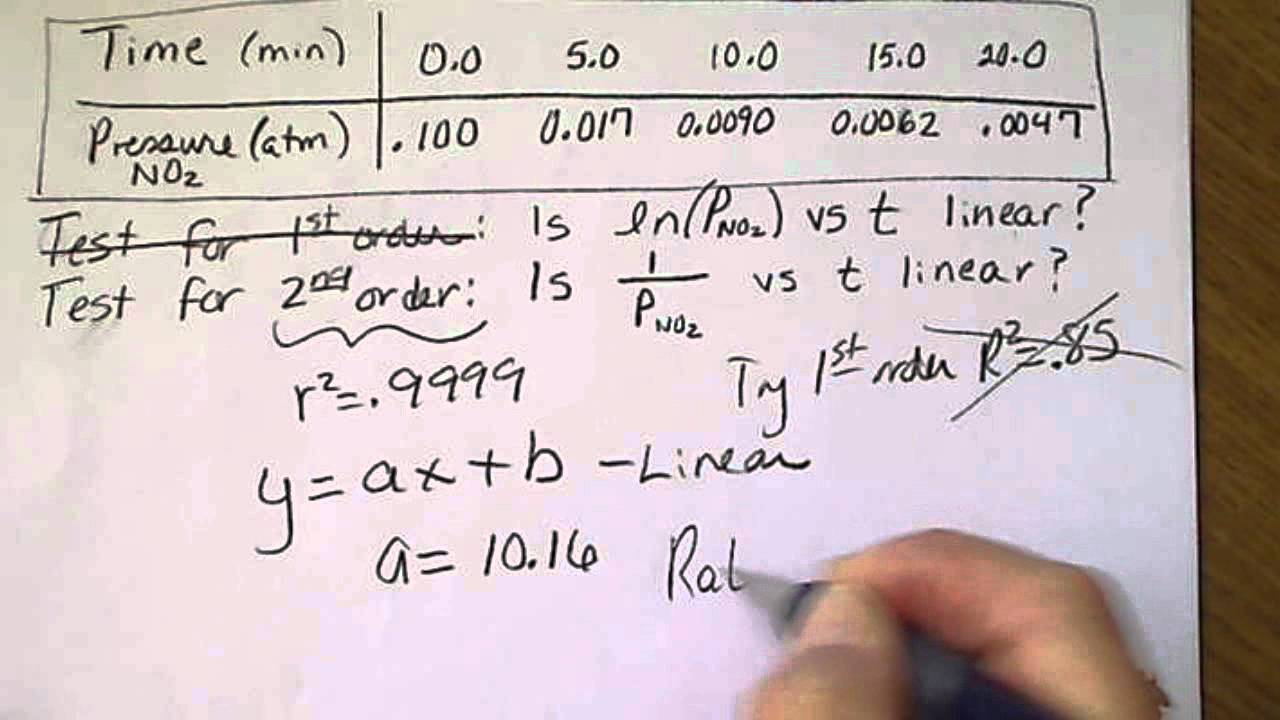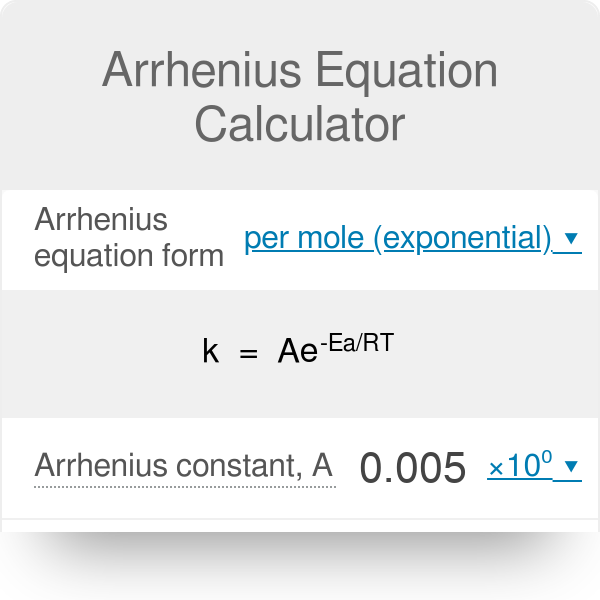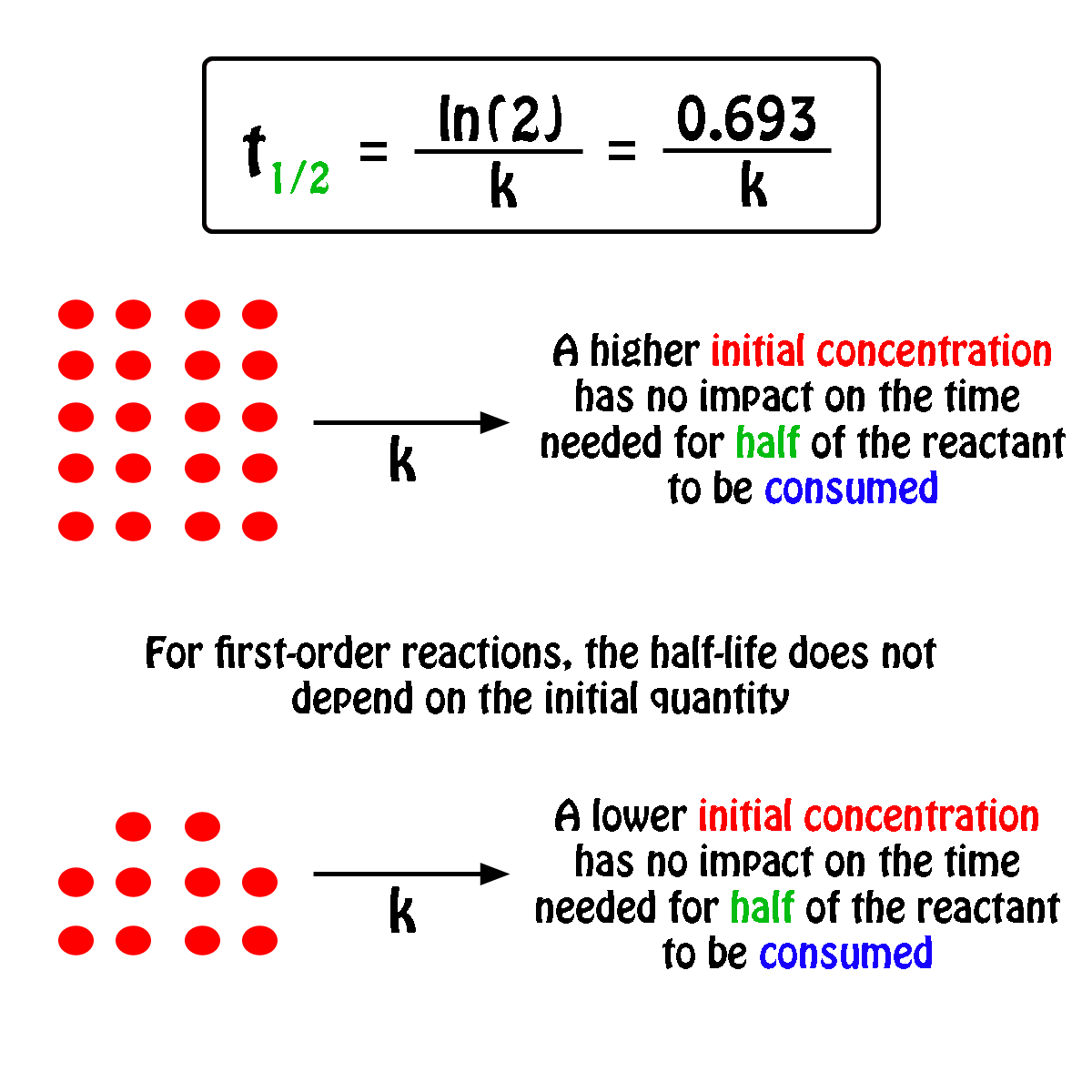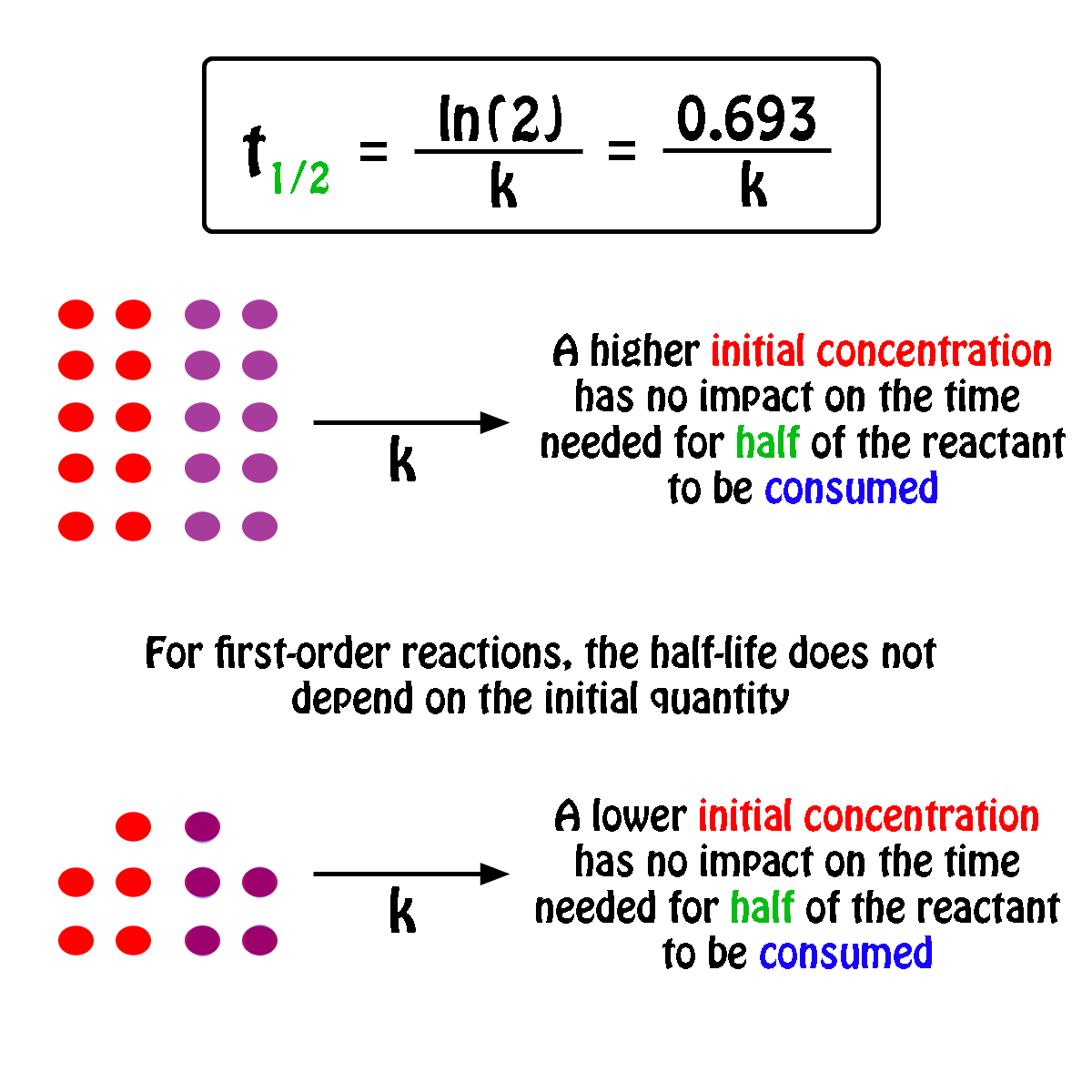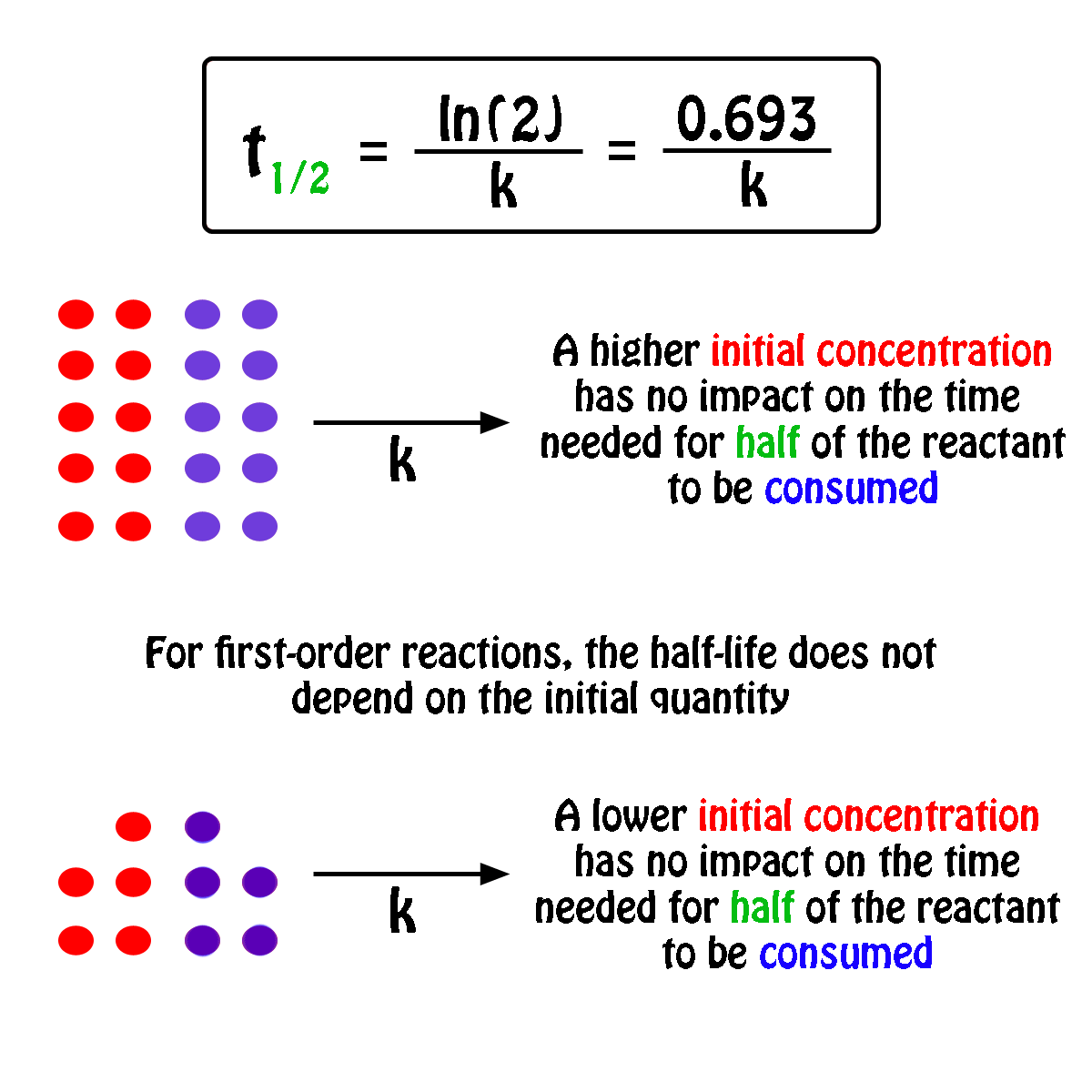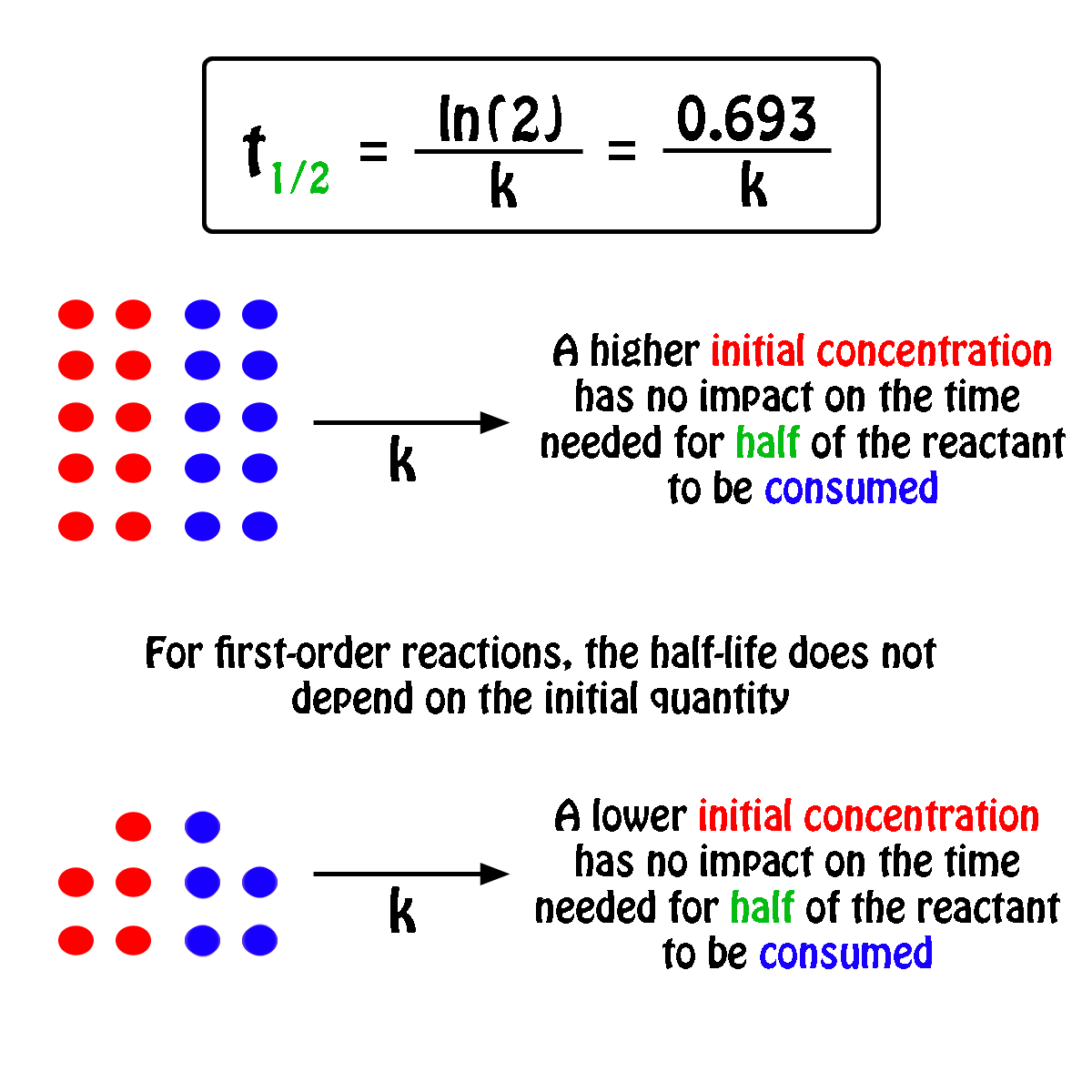
What is the half-life of a first-order reaction with a rate constant of 7.80xx10^-4 s^(-1)? | Socratic
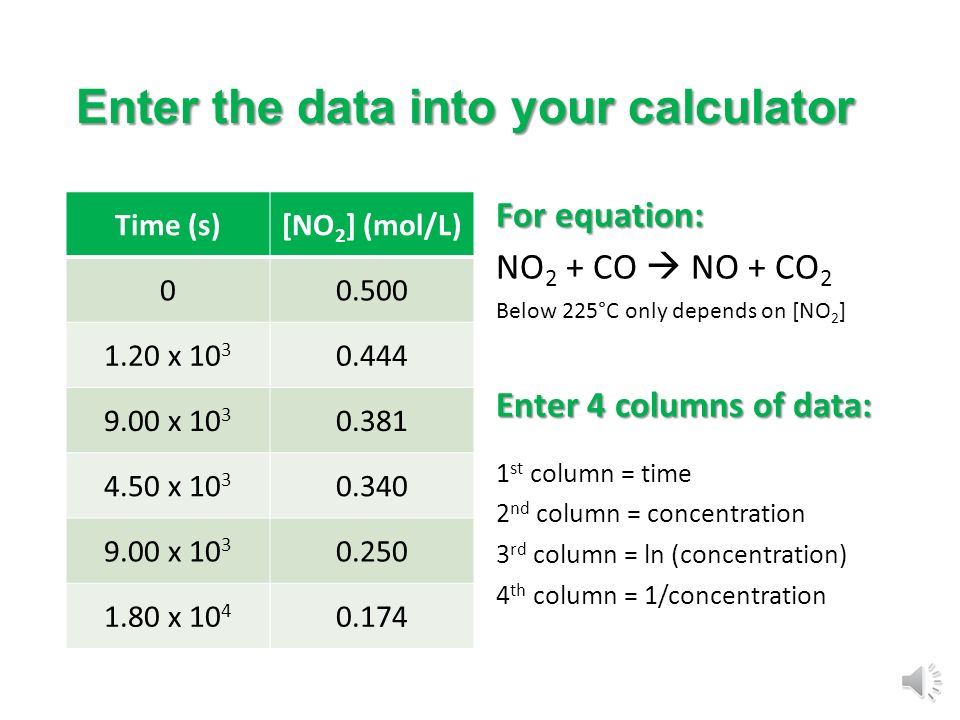
Integrated Rate Law Goal: To determine the order and rate law from concentration and time data. - ppt download

Worked example: Using the first-order integrated rate law and half-life equations | Khan Academy - YouTube

PPT - Summary of the Kinetics of Zero-Order, First-Order and Second-Order Reactions PowerPoint Presentation - ID:545041