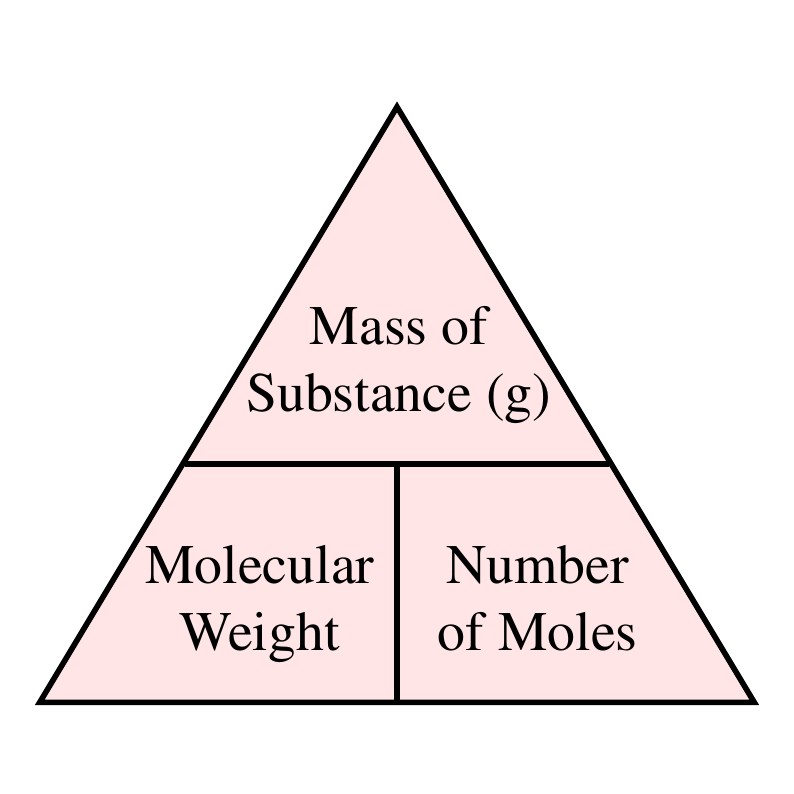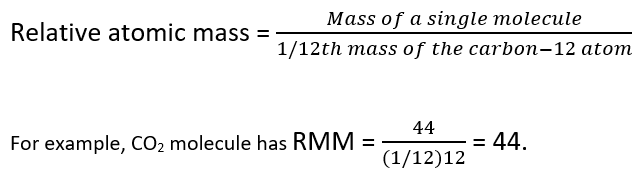
Relative Atomic Mass, Relative Molecular Mass & Mass Spectrometry | A-Level Chemistry Revision Notes

Calculate the percentage of nitrogen in urea NH2CONH2. Given : R.M.M. of N = 14, C = 12 , O = 16, H = 1 ?

How To Calculate Relative Atomic Mass - What is Relative Mass? How to Calculate Relative Atomic Mass along with FAQs

Calculate the number of moles in the following. (A) 160 g of an oxygen molecule (B) 68 g of an ammonia


:fill(ffffff)/https://static-ph.zacdn.com/p/sharp-calculators-0045-8493581-2.jpg)

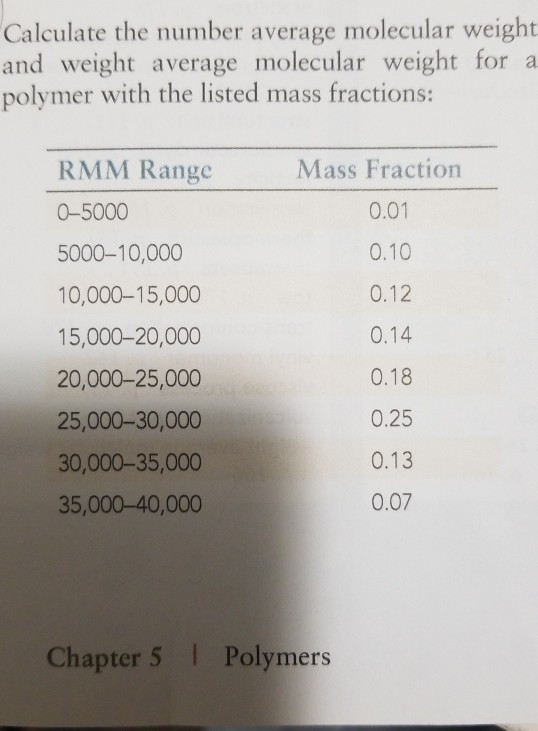








:fill(ffffff)/https://static-ph.zacdn.com/p/sharp-calculators-9514-8693581-1.jpg)